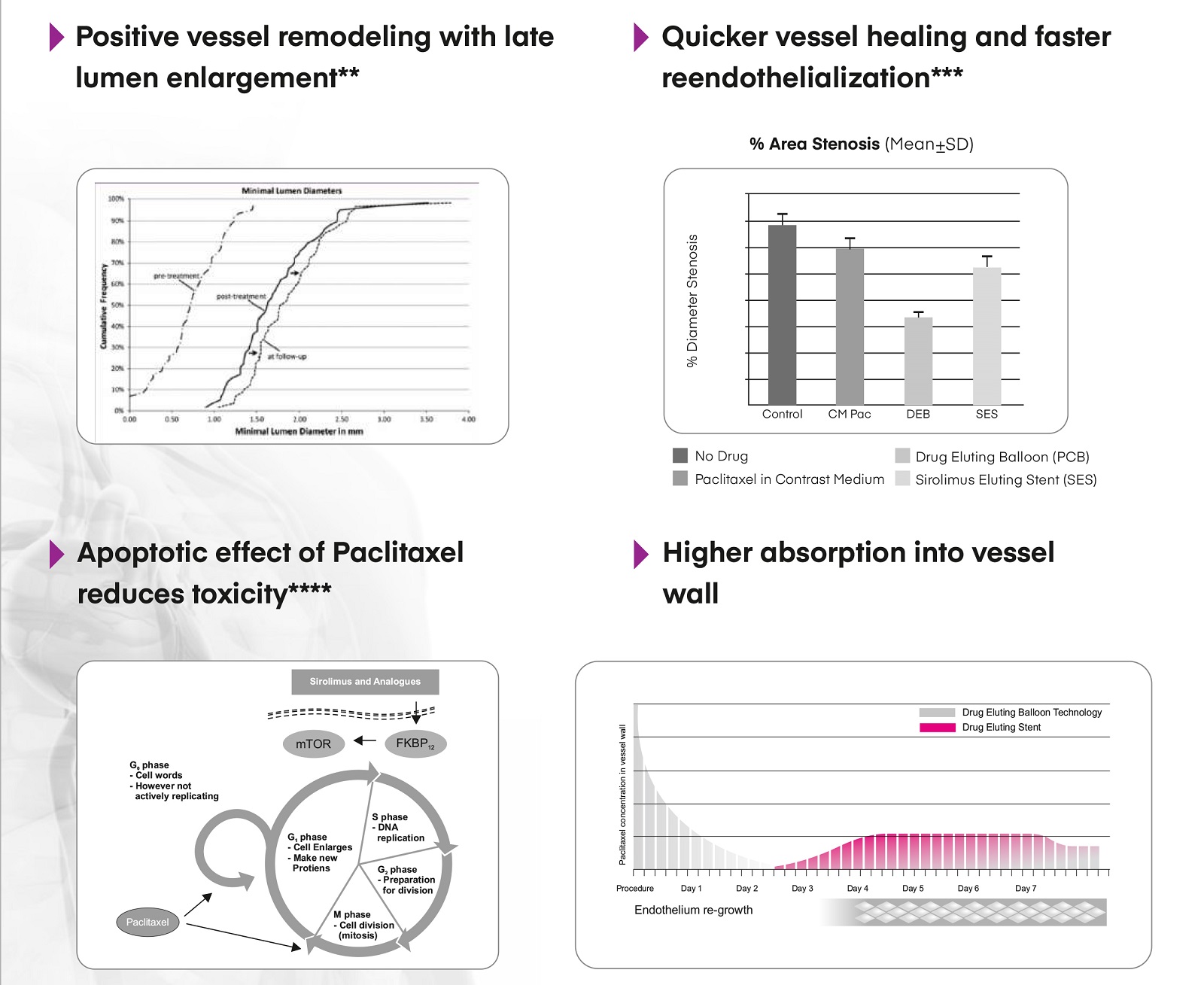
PACLITAXEL – Drug of Choice for DCB in Coronary Interventions with Large Clinical Evidence*
*European Heart Journal (2018)00, 1-23 Doi:10.1093/eurheartj/ehy394 **Clin Res Cardiol. 2015; 104: 217-25
***Speck, U., et al. (2006). “Neointima Inhibition: Comparison of Non–Stent-based Local Drug Delivery and a Drug-eluting Stent in Porcine Coronary Arteries.” RSNA.
****Biotechnology and Bioprocess Engineering 17: 912-924 (2012) DOI 10.1007/s12257-011-0571-z,
Scheller B, Vukadinovic D, Jeger R, et al. Survival After Coronary Revascularization With Paclitaxel-Coated Balloons. J Am Coll Cardiol. 2020;75(9):1017-1028.
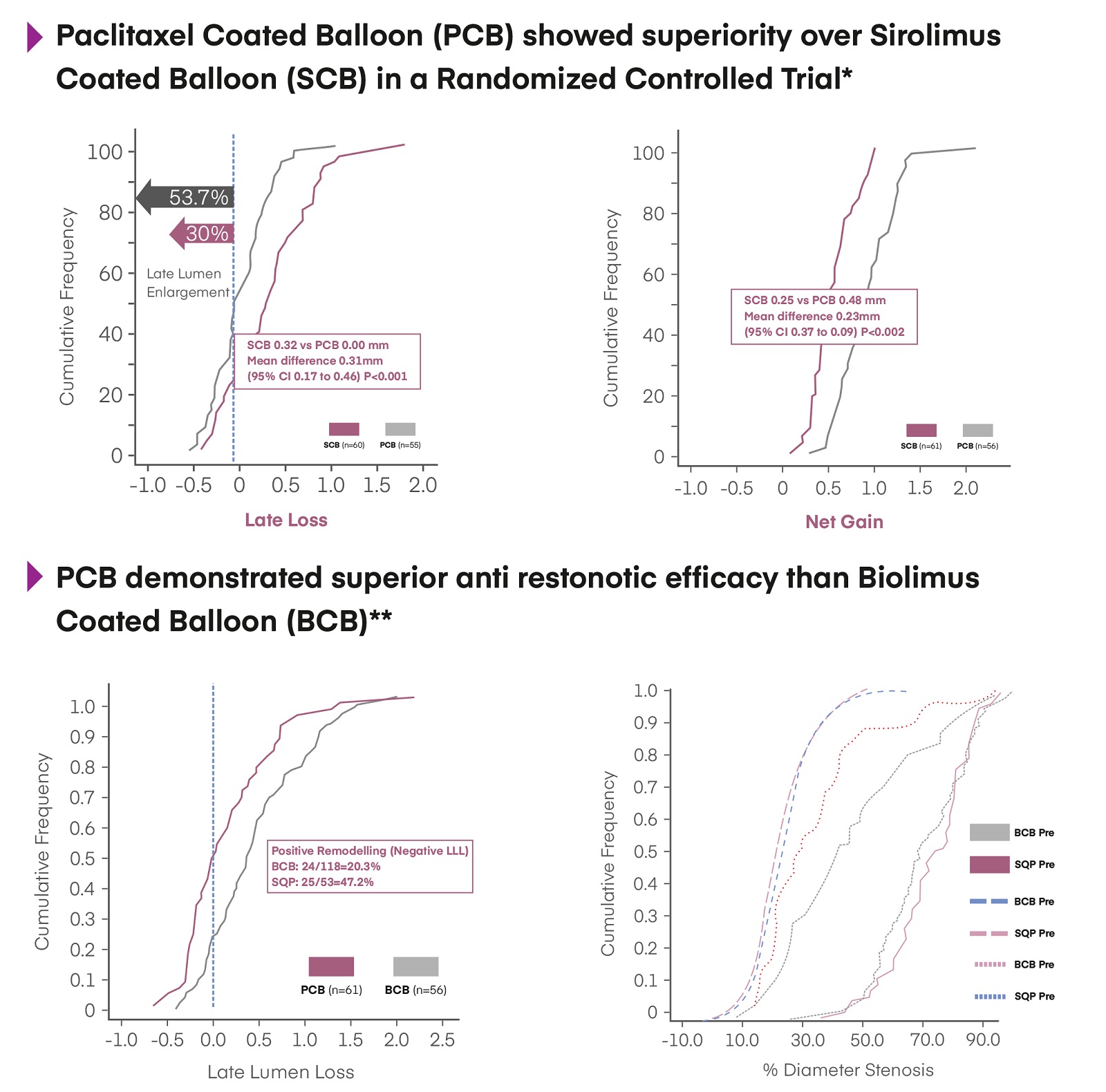
Paclitaxel proven more Efficacious than Limus as a choice of Drug in DCB
*Ninomiya, K. et al. (2023). Sirolimus-Coated vs. Paclitaxel-Coated Balloon in Small Vessels. J Am Coll Cardiol Intv, 16(23), 2884–2896. doi:10.1016/j.jcin.2023.09.026
**Raynor, B. P. et al. (2023). Biolimus A9™ DCB for In-Stent Restenosis: REFORM Trial Design. Cardiovasc Revasc Med, 56, 75–81. doi:10.1016/j.carrev.2023.06.004
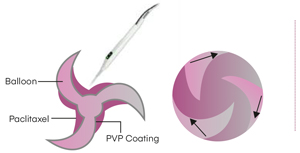
UNIQUE DRUG APPLICATION
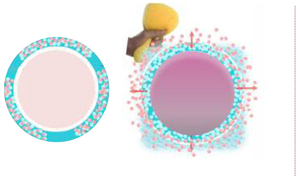
DRUG RELEASE

WING SEAL TECHNOLOGY
- Protects the drug between the balloon folds during advancement, resulting in negligible drug loss prior to inflation
- The corrugation increases flexibility of the balloon ensuring better trackability & crossability
- Tight wrapping prevents the balloon unfolding during advancement
M3i TECHNOLOGY
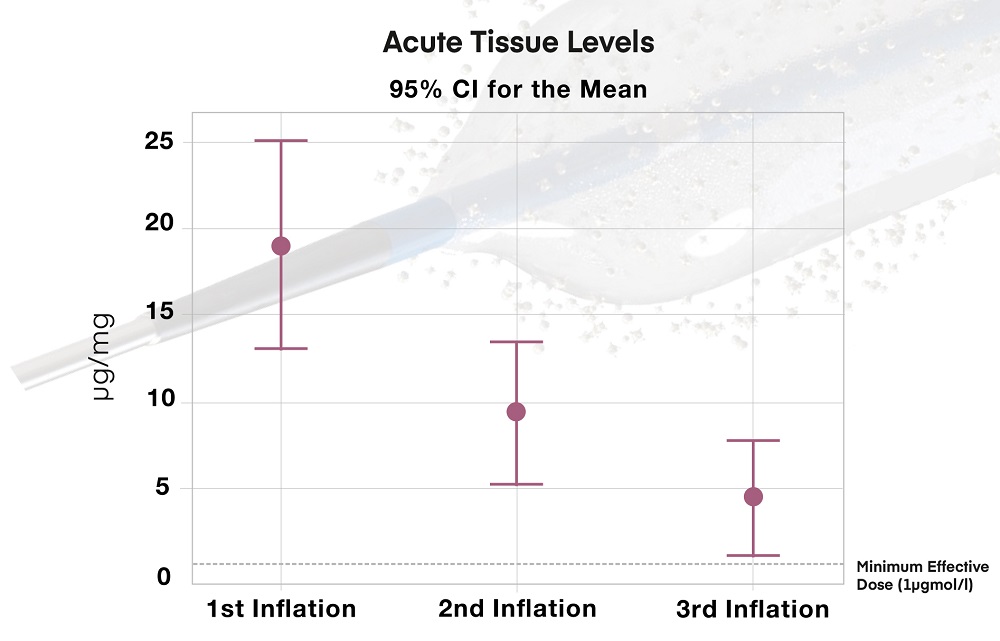
- Multiple Drug Release*
Drug release up to 3 times with the same device when clinically indicated - Interval Plot of Acute tissue levels of Paclitaxel over multiple inflations